
このテンプレートを使用してください
A corrective action plan turns a problem into a solution that sticks. With Trupeer, you can save hours on quality documentation by starting with a free corrective action plan template, customizing it with your brand guidelines, and turning CAPs into video walkthroughs that drive accountability across teams.
This corrective action plan template provides a structured framework to capture issue description, root cause analysis (5 Whys, fishbone), corrective actions, owners, deadlines and verification steps - aligned with CAPA, ISO 9001 and quality management standards. Pair it with our AI SOP creator to update procedures, generate an AI video walkthrough, and translate into 65+ languages for global teams.
How to customize this template in Trupeer
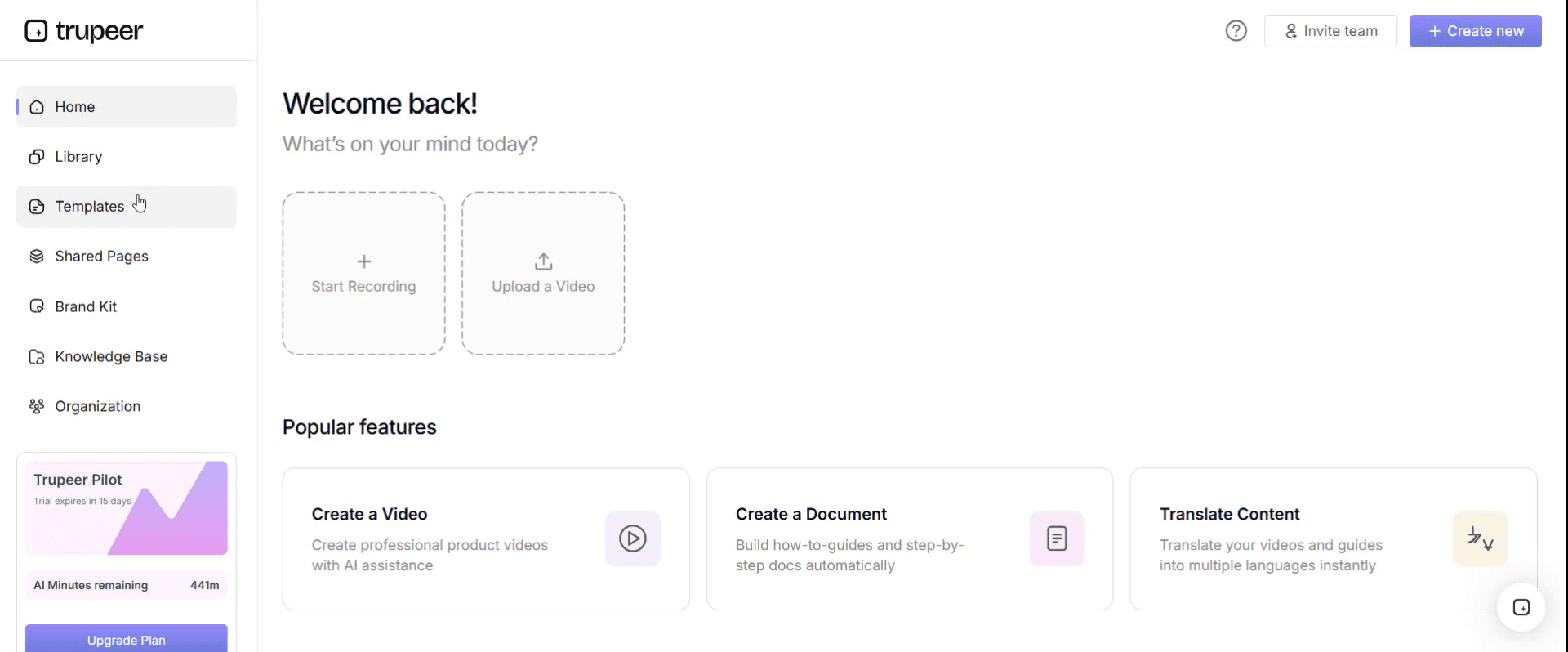
Step 1: Open the Templates Section
Go to the Templates section from the main navigation.
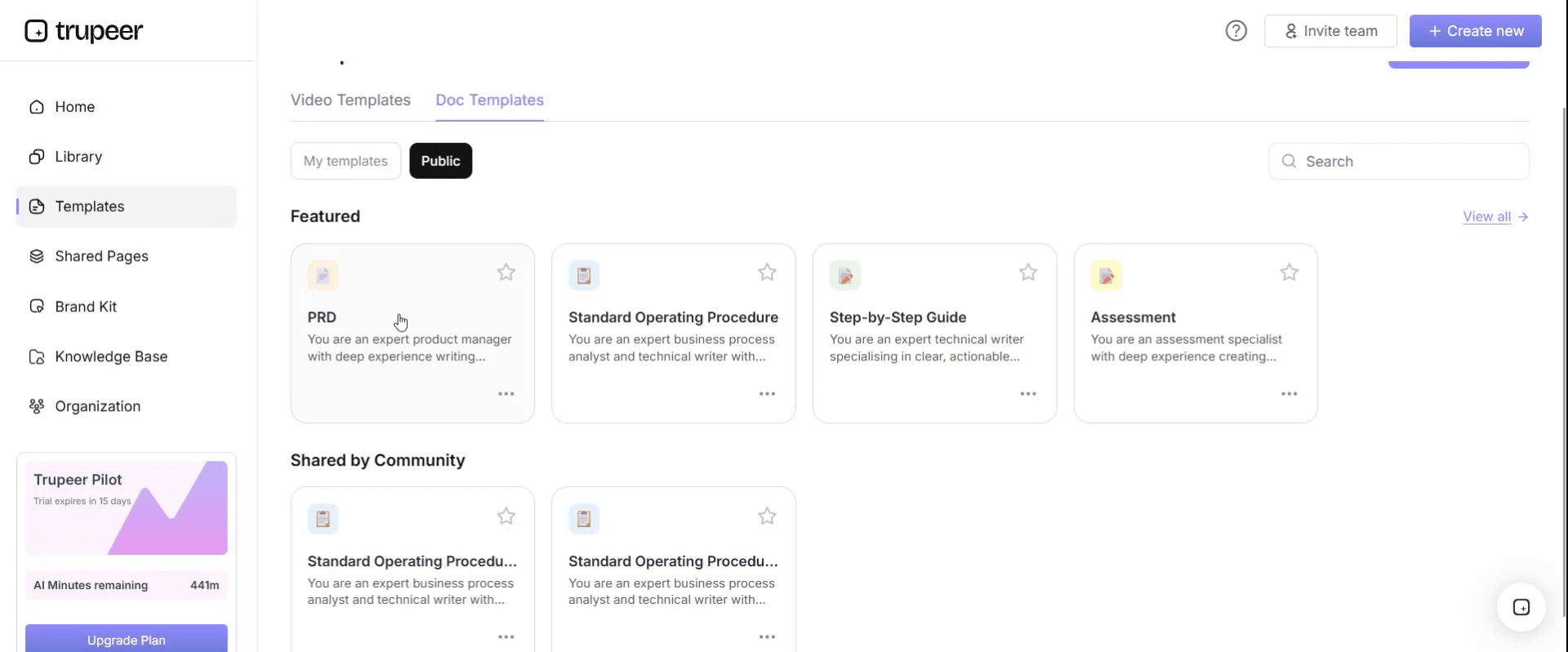
Step 2: Select and Open a Template
Click on any template you want to work with to open it.
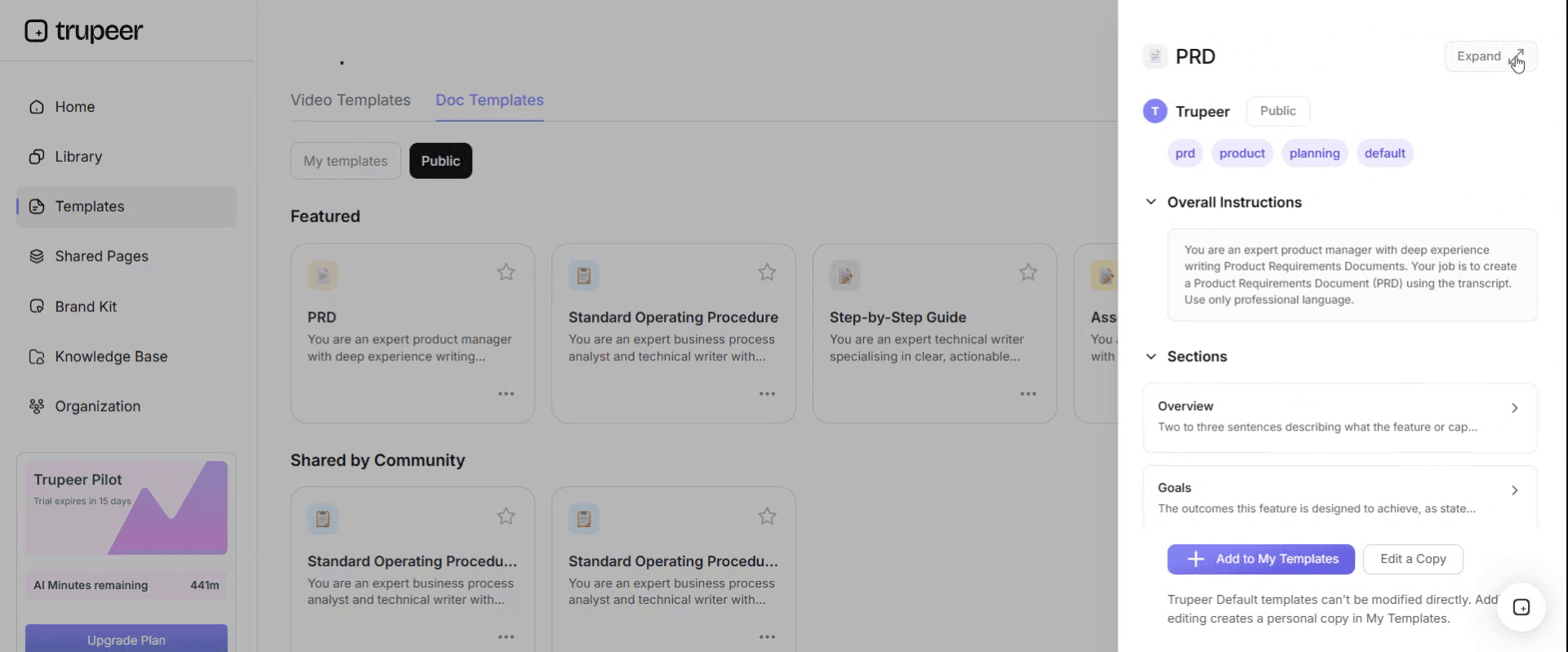
Step 3: Expand the Template View
If needed, expand the template view to see the full layout and details clearly.
Step 4: Edit the Template
Click on Edit to start modifying the selected template.
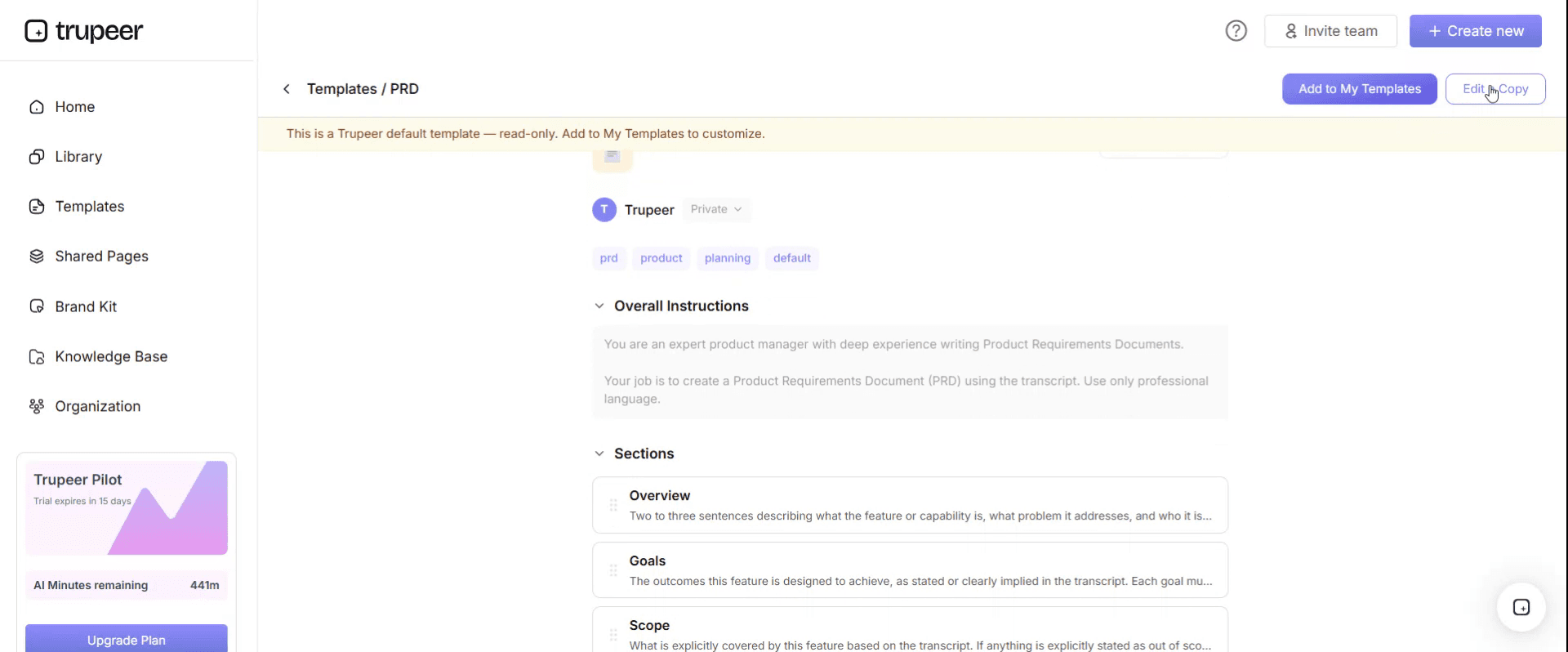
Within the editor, you can:
Add new sections
Define or update formatting rules
Add a logo and adjust its position and related settings
Step 5: Save Your Customized Template
After making all necessary changes, click Save to store the updated template as your own.
Step 6: Preview and Fine-Tune the Template
When you want to see how your customized template looks, open the Preview.
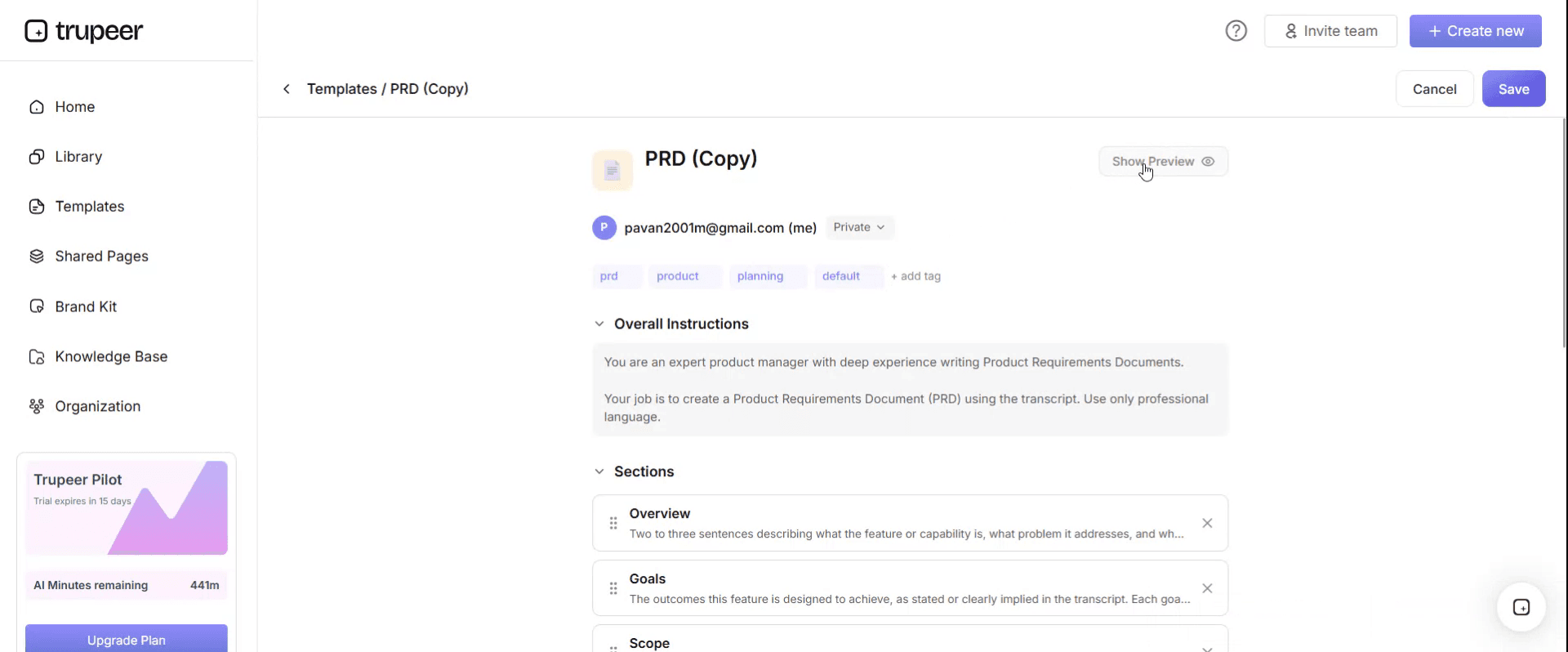
From the preview screen, you can continue to make adjustments directly if needed, ensuring the template appears exactly as you want.
With a corrective action plan template you can:
Save hours on documentation: Skip the blank page with a structure built for CAPA workflows.
Solve problems for good: Built-in root cause analysis prevents the same issue from recurring.
Stay on-brand: Apply your logo, fonts and colors using Trupeer's brand kit.
Train teams on changes: Pair CAPs with video walkthroughs to roll out updated procedures.
Stay audit-ready: Aligned with ISO 9001, IATF 16949 and FDA CAPA expectations.
Reach global teams: Translate CAPs into 65+ languages with one click.
A great corrective action plan stops small issues from becoming big ones. Use this template to capture every CAP clearly, completely and on-brand.
Frequently Asked Questions
What is a corrective action plan?
A corrective action plan (CAP) is a structured document that addresses a problem at its root cause - not just its symptoms. It captures the issue, root cause analysis, corrective actions, owners, deadlines and verification steps to ensure the problem is permanently solved.
What should a corrective action plan include?
A complete CAP includes the issue description, impact, immediate containment actions, root cause analysis (using 5 Whys, fishbone or similar), corrective and preventive actions, owners, deadlines, verification methods, effectiveness check and approval signatures.
What is the difference between corrective and preventive action?
A corrective action addresses an issue that has already occurred - it fixes the root cause to prevent recurrence. A preventive action addresses a potential issue that hasn't happened yet - it removes the risk before it becomes a problem. Together they form CAPA: Corrective and Preventive Action.
How do you write a corrective action plan?
Start by clearly defining the problem. Conduct a thorough root cause analysis - don't stop at symptoms. Define corrective actions that address each root cause. Assign owners and deadlines. Define how you'll verify the actions worked. Sign off and track to closure.
What is CAPA?
CAPA stands for Corrective and Preventive Action. It's a structured quality management process used in regulated industries (pharma, medical devices, automotive) to address non-conformities. CAPA is required by ISO 9001, IATF 16949, FDA 21 CFR 820 and many other quality standards.
